Medical
Medical
Zeus delivers advanced polymer components designed to help medical device engineers access the most challenging and delicate areas of the body with precision and confidence. Leveraging decades of polymer science and extrusion expertise, our team collaborates with you to develop application-specific solutions that enhance device performance, improve clinical outcomes, and raise the level of patient care. Continuously innovating and partnering with medical professionals and manufacturers has kept us and our partners at the forefront of medical device innovation.
To meet the demands of the ever-evolving medical sector, Zeus has developed solutions for a broad spectrum of applications incorporating the polymer properties that matter most. Our fluoropolymers, nylons, LCP, and other engineered plastics have found their way into almost every medical niche. Offering extensive levels of customization, Zeus biocompatible products can be tailored to almost any use within the body, including implantable or bioabsorbable device components.
Medical Markets
Within the medical sector, Zeus serves several vascular and non-vascular market segments. For the vasculature, our catheter components are used in cardio-, neuro– and peripheral vascular applications. In addition to these areas, Zeus products are used in cardiac rhythm management and electrophysiology (CRM/EP), structural heart, and renal denervation procedures. For those non-vascular associated devices, Zeus catheter componentry is found in procedures including gastrointestinal endoscopy (GI ENDO), surgical robotics, diabetes management, oncology, brachytherapy; ear, nose, and throat (ENT), urology, neuromodulation, as well as drug delivery and cosmetic surgery.
Catheter Componentry
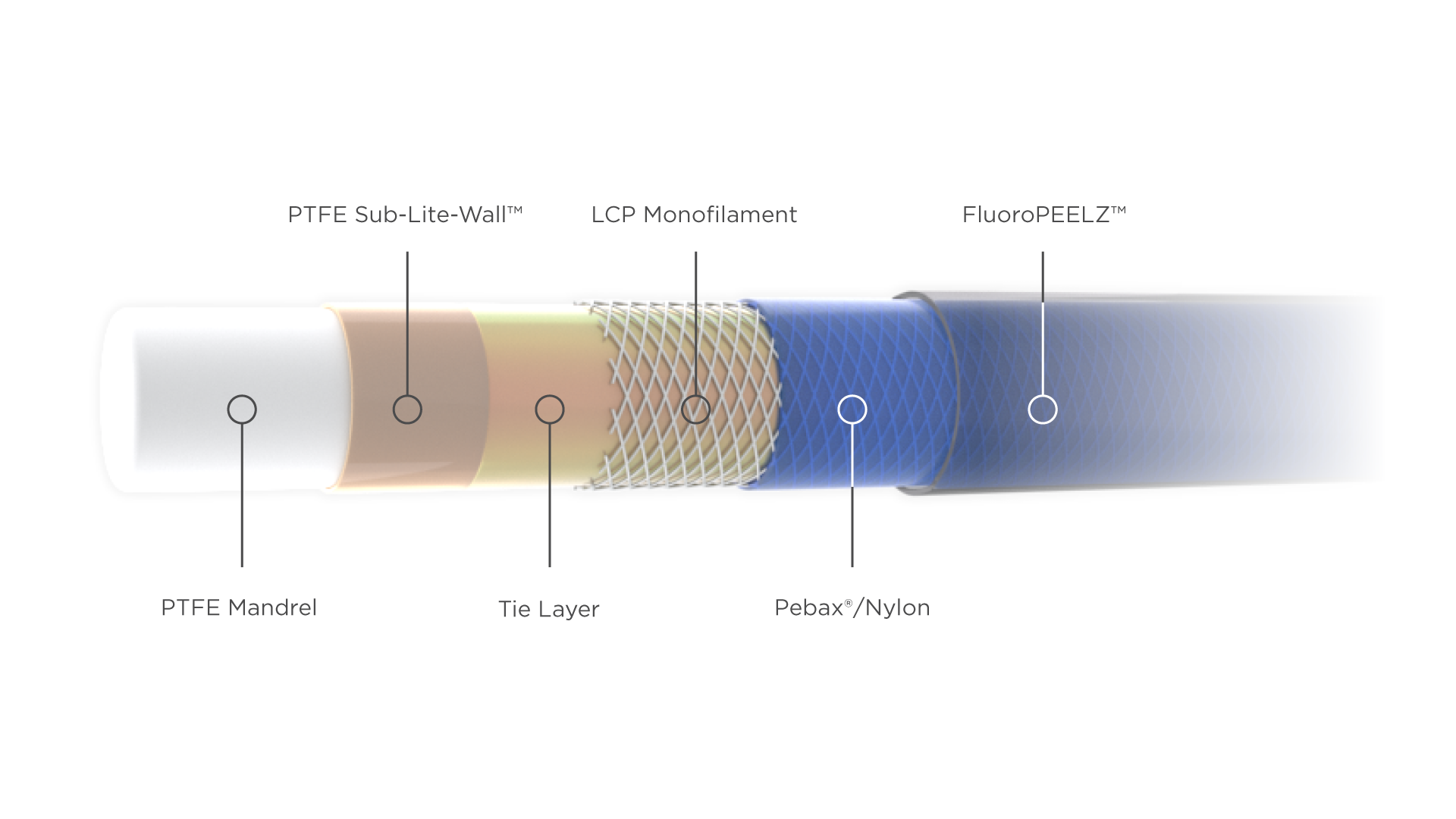
Zeus provides several key components for catheters that are crucial to their end functionality, including torquability, kink resistance, and deflectability. Our PTFE monofilaments are used for mandrels upon which the catheter is made, while Zeus PTFE Sub-Lite-Wall™ or StreamLiner™ Series catheter liners cover the mandrel and are etched to enable bonding to Pebax® or nylon jacketing. Zeus FluoroPEELZ™ peelable heat shrink completes the construction and provides a safe and repeatable means for removing the heat shrink after jacket reflow.
EMERGING PRODUCTS and TECHNOLOGIES
Absorv™ Bioabsorbable Extrusions
Zeus Absorv™ products can safely exist in the body for controlled lengths of time. These customized extruded device components can be made into vascular scaffolds, and can later have coatings applied for use in drug delivery. In addition to standard tubing, Absorv™ can be created in oriented tubing, drawn fiber, monofilament, and ribbon.
Aeos™ ePTFE
Our Aeos™ ePTFE (expanded PTFE) is made using Zeus’ extrusion and expansion processing to create a product with microporous properties. The expansion process creates a porous structure between nodes and fibrils, resulting in a material with microporous and flexible properties highly suited to a number of medical applications. Aeos™ ePTFE can be made into membranes, ribbon, tubing, monofilament (including sutures and high strength sutures), special profiles, and laminates. Membrane and tubing can be biaxially and uniaxially oriented.
Bioweb™ Electrospun Nonwovens
Also made from PTFE, Zeus Bioweb™ nonwovens have microporous properties similar to Aeos™ ePTFE. These custom composites can be used to create implantable structures such as stent coverings, including balloon-expandable and self-expanding stents, and mimic the extracellular matrix to speed healing. Bioweb™ novel encapsulations allow low-temperature encapsulation of stents, preventing struts from contacting the luminal wall and thus reducing their influence on the healing process.
Custom Polymer Solutions for Medical Markets
At Zeus, our polymer scientists, engineers, and technical account managers are ready to assist with all your project needs. Customized solutions are available via phone, e-mail, or video conferencing. In-person consultations are also available on the Zeus campus, or we will come to you.
Contact us today or call toll-free in the US 1-800-526-3842 to learn more about these polymer solutions or any of our other medical device products. Internationally, you can reach us at +1-803-268-9500.